29 January 2024
Gene treatments are being credited with permitting several children born with deafness to hear again.
Gene therapy is a medical treatment that aims to change a few of a patient's genes to cure a genetic disease.
A small study recently published in The Lancet reported improvements in the hearing in five of six children who were treated in China.
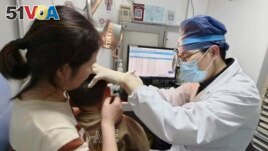
In this photo provided by researchers in January 2024, Dr. Yilai Shu examines a young patient at the Eye & ENT Hospital of Fudan University in Shanghai, China, after a gene therapy procedure for hereditary deafness. (Courtesy Dr. Yilai Shu via AP)
Around the same time, the Children's Hospital of Philadelphia in the U.S. eastern state of Pennsylvania announced similar improvements in an 11-year-old boy treated there. And earlier, Chinese researchers published a study showing similar improvements in two other children.
The experimental methods, or therapies, target only one rare condition. But scientists say similar treatments could someday help many more children with other kinds of deafness caused by genes.
Around the world, 34 million children have deafness or hearing loss. And information from the U.S. Centers for Disease Control and Prevention says genes are responsible for up to 60 percent of cases.
Deafness caused by genes is passed down from parents to children. It is called hereditary deafness. It is the latest condition scientists are targeting with gene therapy. Gene therapy is already approved to treat illnesses such as sickle cell disease and severe hemophilia.
Children with hereditary deafness often get a device called a cochlear implant that helps them hear sound.
"No treatment could reverse hearing loss...That's why we were always trying to develop a therapy," said Zheng-Yi Chen of Boston's Mass Eye and Ear, a treatment center for eye and ear problems. He is a writer of the study published in The Lancet. "We couldn't be more happy or excited about the results."
Treatment results
The team recorded videos of patients. One shows a baby, who formerly could not hear at all, looking back in response to a doctor's words six weeks after treatment. Another shows a little girl 13 weeks after treatment repeating father, mother, grandmother, sister, and "I love you."
All the children in the experiments have a condition that is believed to cause two to eight percent of hereditary deafness. It is caused by changes in a gene responsible for an inner ear protein called otoferlin, which helps hair cells send sound signals to the brain.
The researchers say that the one-time therapy gives a working copy of that gene to the inner ear during a medical operation. Most of the children were treated in one ear, although one child in the two-person study was treated in both ears.
The study with six children took place at Fudan University in Shanghai. Dr. Yilai Shu helped lead the study and trained in Chen's laboratory. Chen was involved in the research. Chinese science organizations and biotechnology company Shanghai Refreshgene Therapeutics helped provide financial support.
Researchers observed the children for about six months. They do not know why the treatment did not work in one of them. But the five others, who were completely deaf, can now hear a normal discussion, the researchers said.
Chen estimated they now hear at a level 60 percent to 70 percent of normal. The therapy caused no major side effects.
Early results from other research have shown similar results.
Regeneron Pharmaceuticals is a biotech company based in New York state. It announced in October that a child under two years old showed improvements six weeks after gene therapy. The results came from a study Regeneron did with support from Decibel Therapeutics, a company in Boston.
Columbia University's Dr. Lawrence Lustig is involved in the Regeneron study. He said although the children in these studies do not end up with very good hearing, "even a moderate hearing loss recovery in these kids is pretty astounding."
He added that many questions remain. They include how long the therapies will last and if hearing will continue to improve in the children.
Some people question if gene therapy for deafness is ethical.
Teresa Blankmeyer Burke is a professor who is deaf and who deals with medical ethics. She teaches at Gallaudet University, a university for deaf people in Washington, D.C. She said that there is no agreement about the need for gene therapy targeting deafness.
She also pointed out that deafness does not cause severe or deadly illness. Blankmeyer Burke said that it is important to work with deaf community members about the importance of gene therapy. She added gene therapy is seen by many as a possible threat to "signing Deaf communities."
However, Chen said: "This is real proof showing gene therapy is working." And he added, "It opens up the whole field."
I'm Faith Pirlo. And I'm Gregory Stachel.
Laura Ungar reported this story for The Associated Press. Gregory Stachel adapted it for VOA Learning English.
______
Words in This Story
hereditary –adj. passed down from one generation to the next
implant –n. (medical) something connected to a person's body through a medical operation
reverse – v. to change (something) to an opposite state or condition
excited – adj. very enthusiastic and eager about something
pretty – adv. to a great degree or extent: very
astounding – adj. causing a feeling of great surprise or wonder
ethical – adj. following accepted rules of behavior: morally right and good












